At first glance, the sheep in the compound of Leipzig's faculty of veterinary medicine look like your ordinary sheep. But if you take a closer look, you see that parts of them have been sheared.
"We took cells from iliac crest and we can't run the risk of contamination," explained Johannes Boltze, head of the Department of Cell Therapy at Fraunhofer Institute for Cell Therapy and Immunology in Leipzig.
The sheep's bone marrow contains stem cells that can transform into a variety of types of cells and which form the basis for the new therapy researchers are putting to the test.
Scientists in Leipzig have already triggered a stroke in some of the sheep while others still await their fate. It's impossible to tell at first glance which of the sheep sufferedd a stroke.
"Just a couple days after the stroke, they don't feel the difference anymore," Boltze said. "The sheep is not impaired in its daily life."
Certain tests, however, can document the subsequent damage. The scientists examine their sheep in a nuclear spin and a positron emission tomography (PET).
And they run different behavioral tests, because the sheep's course of motions is impaired after a stroke - just as it is in humans. The scientists pull away one of the sheep's forelegs. A healthy sheep immediately corrects its stance in order to stand on all fours again. But sheep that had a stroke show a delayed response or don't react at all.
Can stem cells alleviate damages caused by a stroke?
Testing of the researchers' treatment began on lab rats and has been extended to sheep. Shortly after a stroke, the researchers give the animal an injection of immune cells and stem cells. Rats without the injection were not able to balance on a small bar whereas treated rats ran back and forth without a problem.
Sheep serve as substantially better testing grounds. But the quite positive results at the beginning have become ever more infrequent since the scientists started to conduct blind studies in which scientists do not know whether a sheep was treated with a stem cell injection.
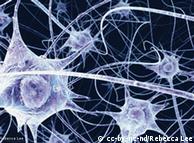 About 30,000 cells can die each second when blood vessels to the brain are clogged
About 30,000 cells can die each second when blood vessels to the brain are clogged"None of the scientists was deliberately altering the results, but your own expectations do have an impact," said Boltze. Now, the positive results have decreased, but the scientists hope that the remaining results are even more significant and can be easily transferred to the human body.
Currently, tests have called for injecting stem cells of the same animal. The next step will be to transfer human cells into the sheep. At one of Fraunhofer's laboratories, Alexandra Stolzing tries to recover stem cells from human skin. The difficultly is that skin cells do not naturally have the ability to form other types of cells. In order to work as stem cells, they need to be forced to "forget" the transformation they underwent to become a specific cell type. They need to be reset to be able to transform into another type of cell, such as a nerve cell.
The reprogramming is a complicated process that is affected by a number of factors, including the age and condition of a cell and the medium in which the process is due to take place. The researchers have just begun to understand the details. It's an exciting process that could go beyond being beneficial for stroke research.
"I work in the field of ageing research and it's fascinating to me that a cell can be brought back to a state that can be considered young," Stolzing said.
Still a long way to go
The scientists are still working to understand exactly what influence the stem cells have on the body. They know so far that the cells don't wander to the brain, build new neurons and replace the impaired tissue. This process would take at least four weeks, but the positive results of the stem cell injection shows after about a week, said Boltze.
"We assume that the cells block certain secondary complications, affect inflammation and minimize cell death," Boltze said, adding that stem cell therapy could become an important part of stroke treatment plans. "Maybe in total the result will be slightly better as without the stem cells."
The key is still how fast the patient reaches a proper clinic so that the blood clot in the brain can quickly be dissolved. Boltze consistently gets requests from patients that he cannot meet both for ethical and medical reasons. Since the methods are still in animal trials, Boltze cannot use them on human patients, and there is no established way to transfer stem cells to humans within the short three-day window after a stroke that the tests require.
Scientists in the United States have already started the first clinical trials and others are set to begin in Germany in 2012. But even if all trials are successful, in will take at least 10 years until the stem cell therapy will be available. In any case, preventing a stroke is better than treating one, said Boltze.
"Medicines are not the best solution," Boltze said. "The best solution is a healthy lifestyle. Don't smoke, drink only a little alcohol and exercise. That's more beneficial than any stem cell."
Author: Claudia Ruby / sst
Editor: Sean Sinico


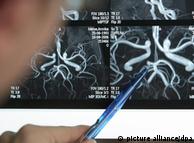

 About 30,000 cells can die each second when blood vessels to the brain are clogged
About 30,000 cells can die each second when blood vessels to the brain are clogged


 In many parts of the world, the beginning of November marks the start of a month-long series of activities aimed at raising diabetes awareness and calling for urgent action to tackle the diabetes epidemic. Here's a reminder of why we all have to 'Act on Diabetes. Now.':
In many parts of the world, the beginning of November marks the start of a month-long series of activities aimed at raising diabetes awareness and calling for urgent action to tackle the diabetes epidemic. Here's a reminder of why we all have to 'Act on Diabetes. Now.':